EConsent in Healthcare Market Share Analysis Across Hospitals and Research Organizations
Global EConsent in Healthcare Market: Digital Consent Transforming Patient-Centric Healthcare
Market Overview
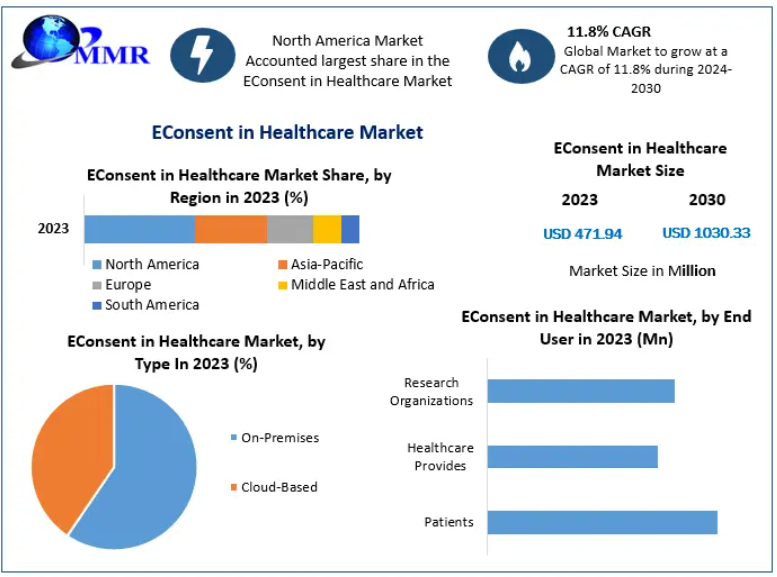
The global EConsent in Healthcare Market was valued at USD 471.94 million in 2023 and is projected to reach USD 1,030.33 million by 2030, growing at a CAGR of 11.8% during the forecast period. The market is expanding steadily as healthcare organizations, clinical research institutions, and pharmaceutical companies adopt digital solutions to streamline informed consent processes and improve patient engagement.
EConsent refers to the use of electronic platforms to obtain informed consent for clinical trials, medical procedures, telehealth services, and healthcare data sharing. By replacing traditional paper-based consent forms with interactive digital systems, eConsent enhances accessibility, improves comprehension, and supports regulatory compliance.
To know about the Research Methodology :- Request Free Sample Report@https://www.maximizemarketresearch.com/request-sample/219294/
Digital Transformation Reshaping Informed Consent
The healthcare sector is rapidly shifting toward digital workflows, and eConsent has emerged as a key innovation in modern patient communication. Traditional consent processes often rely on lengthy paper documents that can be difficult for patients to understand and cumbersome for providers to manage.
EConsent systems solve these challenges by delivering information through multimedia formats such as videos, graphics, interactive documents, and mobile applications. Patients can review consent materials on smartphones, tablets, or computers, making the process more convenient and transparent.
Key Market Drivers
Growing Adoption in Clinical Trials
Clinical trials represent one of the largest application areas for eConsent solutions. Research organizations are increasingly adopting digital consent platforms to improve participant enrollment, reduce administrative burden, and enhance compliance with regulatory standards.
EConsent provides trial participants with a more engaging and understandable consent experience, helping improve recruitment rates and participant retention.
Government Support and Regulatory Compliance
Government agencies across major healthcare markets are supporting digital transformation in healthcare, including electronic consent solutions. Regulatory frameworks focused on data security and patient privacy are encouraging healthcare providers to implement secure consent systems.
In regions such as United States and European Union, compliance requirements such as HIPAA and GDPR are accelerating the adoption of secure electronic consent platforms.
Expansion of Telehealth and Remote Monitoring
The growth of telehealth and remote patient monitoring services is creating significant demand for eConsent solutions. As healthcare delivery increasingly shifts to virtual platforms, providers need secure and user-friendly digital tools to obtain patient consent remotely.
Market Challenges
Lack of Awareness and Adoption Barriers
Despite strong growth potential, many healthcare providers and patients still have limited awareness of eConsent technologies. Concerns around data privacy, digital literacy, and system reliability can slow adoption.
Technical Integration Challenges
Integrating eConsent platforms with existing healthcare IT systems such as electronic health records and clinical trial management software can be technically complex. Compatibility issues and implementation costs remain important barriers.
Segment Analysis
By Type
The market is segmented into:
- On-Premises
- Cloud-Based
The Cloud-Based segment is expected to grow rapidly due to scalability, remote accessibility, and lower infrastructure costs. Cloud deployment also supports easier updates and broader patient access across healthcare networks.
By Application
The major applications of eConsent in healthcare include:
- Clinical Trials
- Medical Procedures
- Telehealth and Remote Monitoring
- Data Sharing and Health Information Exchange
Among these, Clinical Trials dominate the market due to increasing pharmaceutical research activities and the growing need for streamlined patient recruitment.
By End User
Key end users include:
- Patients
- Healthcare Providers
- Research Organizations
Research organizations remain a major adopter, while hospitals and healthcare providers are rapidly integrating eConsent into routine medical workflows.
To know about the Research Methodology :- Request Free Sample Report@https://www.maximizemarketresearch.com/request-sample/219294/
Regional Insights
North America Leads the Market
North America holds the largest share of the global eConsent in healthcare market due to advanced healthcare digitization, strong regulatory frameworks, and widespread adoption in clinical research.
The United States leads regional growth, driven by large-scale clinical trials, strong health IT infrastructure, and increasing telehealth adoption.
Europe Shows Strong Regulatory-Driven Growth
Europe is witnessing substantial market expansion due to GDPR compliance requirements and growing digital healthcare investments.
Asia Pacific Emerging as High-Growth Region
Asia Pacific is expected to experience rapid growth, supported by healthcare modernization initiatives in China, India, and Japan.
Competitive Landscape
The global eConsent in healthcare market features a competitive mix of established health technology providers and specialized digital trial companies. Major players include:
- Advarra
- Castor
- IQVIA
- Medable
- Signant Health
- Suvoda
- Veeva Systems
- Florence Healthcare
These companies are focusing on mobile-first platforms, multilingual support, secure e-signatures, and integration with broader eClinical ecosystems.
Emerging Trends
Mobile eConsent Applications
Mobile-friendly consent platforms are becoming increasingly popular, allowing patients to review and sign consent forms through smartphones and tablets.
Multimedia Consent Experience
Healthcare organizations are enhancing patient understanding through:
- Embedded videos
- Interactive graphics
- Consent acknowledgment checks
- Digital documentation
These features improve participant comprehension and engagement.
Strategic Partnerships
Collaborations between eClinical software providers are increasing to create unified platforms that combine eConsent with electronic patient-reported outcomes and remote trial management.
Future Outlook
The global eConsent in healthcare market is expected to continue strong growth as healthcare providers and clinical research organizations accelerate digital transformation. The demand for secure, efficient, and patient-friendly consent solutions will increase across clinical trials, telehealth, and routine medical care.
As healthcare systems worldwide prioritize patient engagement, process optimization, and regulatory compliance, eConsent is set to become a foundational component of modern healthcare delivery by 2030.
- Art
- Causes
- Crafts
- Dance
- Drinks
- Film
- Fitness
- Food
- Παιχνίδια
- Gardening
- Health
- Κεντρική Σελίδα
- Literature
- Music
- Networking
- άλλο
- Party
- Religion
- Shopping
- Sports
- Theater
- Wellness



